
My Background
I am a medical doctor trained and certified in Internal Medicine, and in Allergy and Immunology from the US, and also I have a PhD in Immunology from Canada. I have experience in human medical product development, early and late phase clinical trial design and analysis, and regulatory strategy for product approval in the US and globally. I am experienced in scientific and regulatory aspects of small molecule drug and large molecule biologic product development in the clinical areas of respiratory medicine, allergy and immunology, rheumatology, and autoimmune diseases. I have experience in development of medical products for common diseases such as asthma, chronic obstructive pulmonary disease, and rheumatoid arthritis, and less common diseases such as pulmonary fibrosis, cystic fibrosis, and vasculitides. I also have basic science and clinical science expertise to study health harm reduction of smoke-free products compared to combustible cigarettes, and nicotine science broadly.
My Experience
US Food and Drug Administration
1997 to 2018, Various Leadership Positions
Industry Roles
2018 to 2019, AstraZeneca, Senior Vice President
2019 to 2022, Savara Inc., Chief Medical Officer
2022 to 2026, Philips Morris International, Chief Life Science Officer
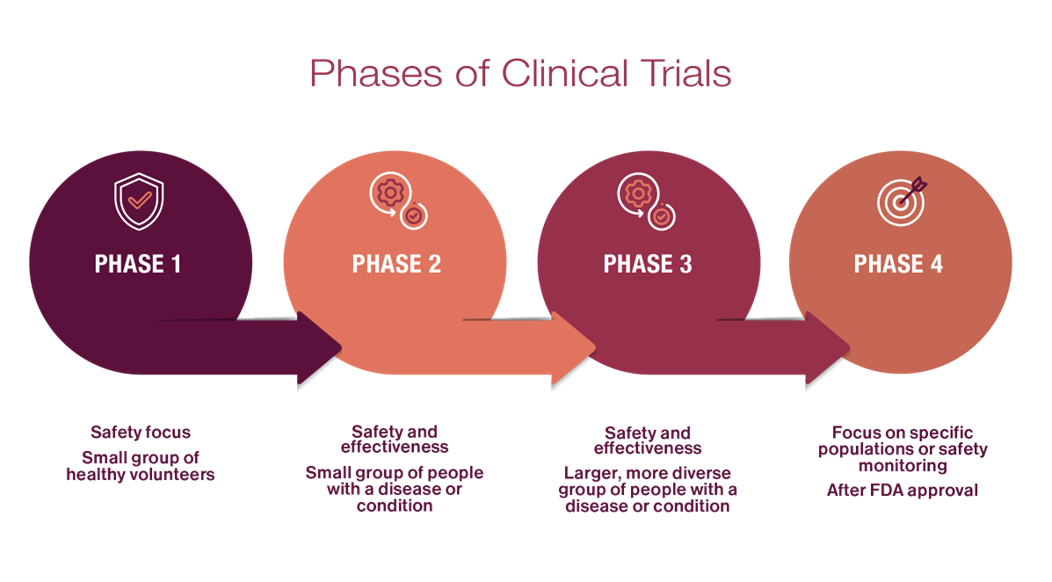
Clinical Trials
Guiding the design and execution of human clinical studies for respiratory diseases, allergy and immunology, rheumatology, and autoimmune diseases.
Regulatory Compliance
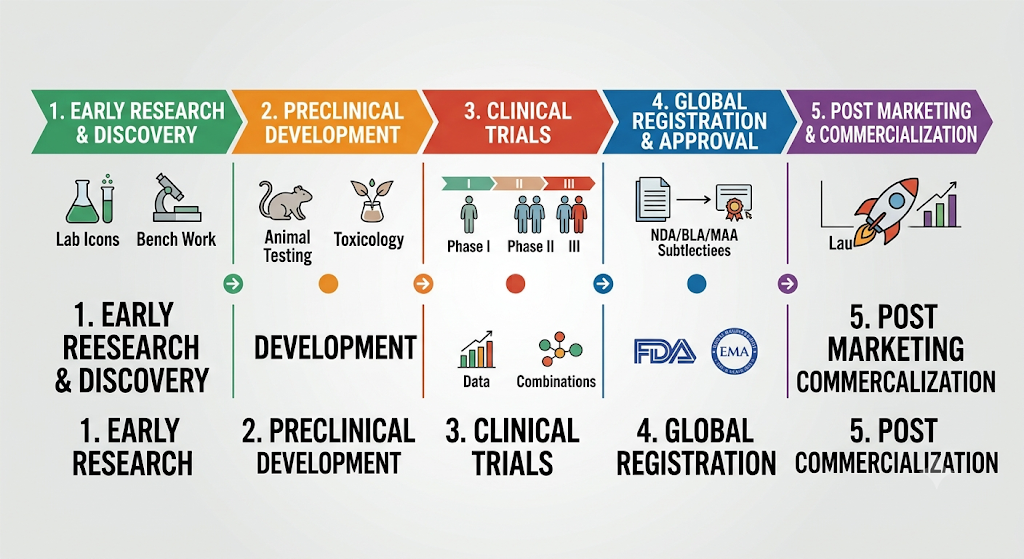
Product Development
BC Pharma Consulting LLC transforms complex ideas with a product into a manageable clinical development strategy, makes complex regulations clear and manageable, and transforms ideas into a marketed product.
